Prostate Cancer and Sexual Function are interwoven. Most of the forms of treatment for prostate cancer can have a significant effect on sexual function. One reason is the fact that the nerves controlling sexual functioning run along the outside of the prostate gland on their way to the penis. In the course of surgery to remove the gland the nerves may get damaged or even cut on one or both sides. With radiotherapy the nerves may get damaged because they are in the path of the radiation to the prostate. Even if a man does not have either surgery or radiotherapy his sexual functioning can be seriously affected by treatments such as Androgen Deprivation Therapy (also known as Hormone Treatment). The hormone treatment reduces libido and seeks to block the normal production of testosterone. When we bear in mind that the majority of prostate cancer patients are diagnosed late in their mid to late 60s their sexual functioning may already have diminished due to lower testosterone or other reasons such as diabetes type 2. The further reduction due to their cancer treatment may be all the more dramatic.
The EUROPROM Study indicates that the level of sexual functioning after treatment is much lower than previous clinical studies of Quality of Life have shown. The study does not tell us why but the anecdotal discussions among patients suggests that in the clinical setting men may not be as comfortable about indicating the true level of their sexual functioning as when they were in the privacy of their own homes when answering the survey.
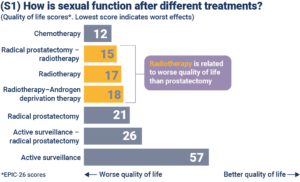
Slide S1 shows quality of life score: the bigger the score the better quality of life. The level of the score relates to sexual function, after different treatments this varies quite a bit, but are below the published clinical studies. In S1 the slide shows that the sexual quality of life was better after prostatectomy than radiotherapy, which may be surprising to some. But the difference of 4 points between radiotherapy and radical prostatectomy is small and may not be clinically relevant.
But quality of life scores for both treatments are obviously low compared with active surveillance.
Why is the active surveillance score not 100, which is the top of the scale in the EPIC Questionnaire score? Clearly it is because at these ages (the average age of the study is 70) sexual function is not normally 100%.
Comparing these figures to the general population, the average EPIC sexual function score for men without prostate cancer is 55.8, which is very similar to the active surveillance score shown here.
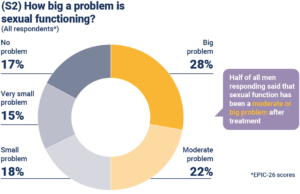
Slide S2: How big a problem then, is sexual functioning as men report it? You can see a big or moderate problem in around half of men. One of the intriguing questions is how these scores would be affected if we were able to ask the patients’ partners the same question.
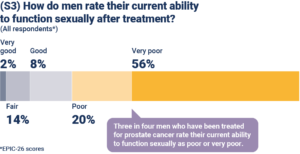
Slide S3: Around three quarters of men who responded to the survey rated their current ability to function sexually as poor or very poor. This is clearly partly a reflection of the age of respondents, as well as the effects of treatment.
However, for comparison, it is interesting to look at a 2017 study of men of slightly older age (average age 74.5) who did not have prostate cancer, which used the same EPIC-26 measures (Venderbos et al, PMID: 28168601). It found that 50% of these men rated their ability to function sexually as poor or very poor. Clearly, this is a significantly lower percentage than the 76% of men with prostate cancer in our study.

Slide 5 Only 34% of men have tried medications and devices to improve erections, so there’s clearly a need to give men more advice on these approaches to help overcome any problems.
It appears that at least some of the men who say they have problems with their sexual function do not appear to do much about it.
In the next issue we will focus on what the survey tells about urinary incontinence.
You can read about our EUPROMS study and download the whole EUPROMS presentation at here.